Dinges, D. F., Orne, M. T., Whitehouse, W. G., & Orne, E. C. Temporal placement of a nap for alertness: Contributions of circadian phase and prior wakefulness. Sleep, 1987, 10(4), 313-329.
David F. Dinges, Martin T. Orne, Wayne G. Whitehouse, and Emily Carota Orne
Unit for Experimental Psychiatry, The Institute of Pennsylvania Hospital, and Department of Psychiatry, University of Pennsylvania School of Medicine, Philadelphia, Pennsylvania, U.S.A.
Summary: Napping can enhance alertness during sustained wakefulness, but the importance of the temporal placement of the nap between days and within the circadian cycle remains controversial. To resolve these issues, a betweengroups study was conducted with 41 healthy, young adults permitted a 2-h nap at one of five times during a 56-h period otherwise devoid of sleep. Naps were placed 12 h apart, near the circadian peak (P) or trough (T), and were preceded by 6, 18, 30, 42, or 54 h of wakefulness. Visual reaction time (RT) performance, Stanford Sleepiness Scale (SSS) ratings, and sublingual temperature were assessed every few hours throughout the 56 h, which took place in an environment free of time cues. All groups displayed a circadian-modulated decline in RT measures and increases in SSS functions as sleep loss progressed. A nap placed at any time in the protocol improved RT performance, particularly in the lapse domain, but not SSS ratings. Comparisons within groups of circadian temperature cycles for the first versus second day of the protocol indicated that early naps (P6, T18, P30) tended to prevent the mean drop in temperature across days. The earlier naps (P6, T18) yielded more robust and longer lasting RT performance benefits, which extended beyond 24 h after the naps, despite the fact that they were comprised of lighter sleep than later naps. Circadian placement of naps (P vs. T) did not affect the results on any parameter. In terms of temporal placement, therefore, napping prior to a night of sleep loss is more important for meeting subsequent performance demands than is the circadian placement of the nap. SSS ratings suggest that the napper is not aware of these performance benefits. Because the longest lasting RT gains followed early naps, which were composed of less deep sleep than later naps, napping during prolonged sleep loss may serve to prevent sleepiness more readily than it permits recovery from it. Key Words: Naps--Alertness--Performance-Sleep deprivation--Circadian phase.
Napping has received much attention as a possible countermeasure to the effects of sleep loss due to sustained activity. A "nap" may be considered as any sleep period
Accepted for publication December 1986.
Address correspondence and reprint requests to Dr. David F. Dinges at Unit for Experimental Psychiatry, Institute of Pennsylvania Hospital, 111 North 49th Street, Philadelphia, PA 19139-2798, U.S.A.
313
314 D. F. DINGES ET AL.
<50% of a person's average nocturnal sleep length. Defined thus, a single nap in a 24-h period otherwise devoid of sleep opportunities would not be considered enough sleep to maintain performance or alertness fully, since humans require at least 50% of their typical nocturnal sleep length to avoid showing performance deficits during acute periods of sustained work (1,2). On the other hand, naps are often alleged to be refreshing disproportionate to their length, and naps distributed throughout a 24-h day have been reported to be an effective countermeasure to performance degradation (3-6), nearly as effective as a single long sleep period (3,5,6). Indeed, numerous studies have demonstrated that in terms of behavioral measures of sleepiness, even "a little sleep," in the form of a single nap in a 24-h period, is better than no sleep at all (7-14) and, generally, the longer the nap the greater the benefits (8,9,13,15). A nap lasting 1-4 h is rarely as beneficial as an 8-12-h sleep, however (13,15-19).
Although some sleep is consistently observed to be better than no sleep at all and the longer the sleep the greater the increase in alertness, a number of issues concerning the benefits of a nap during sustained wakefulness remain controversial. These issues can be divided into two classes of problem: the temporal placement of a nap and the measurement of nap effects. Each of these has important theoretical implications for our understanding of sleep and practical consequences for the use of naps during sustained work scenarios.
The problem of temporal placement of a nap during sustained wakefulness involves two questions. The first concerns the placement of a nap between ›or = 2 days of wakefulness: On which day is a nap most likely to yield the optimum benefits if it is the only sleep taken during ›or = 48 h of wakefulness? Orne (20) suggested that "prophylactic napping," which refers to napping in advance of sleep loss, may be more beneficial than napping after sleep debt accumulation, a hypothesis that seems to contradict the view that sleep cannot be "stored" in anticipation of sleep loss. The issue has not been appropriately tested in most napping studies because the paradigms typically permit a nap only after a period of ›or = 24 h of wakefulness. Those studies that have attended to nap effects in advance of sleep loss (<24 h) or during sleep restriction have yielded data suggesting that an early nap may be advantageous (3,5,7,11,12,18,21), although no assessment contrasting the effects of naps taken prior to and following sleep loss has yet been made.
The second temporal placement issue concerns the timing of a nap within each 24-h cycle of sustained wakefulness. Naitoh (22) reported that a 2-h nap near the circadian nadir in alertness (0400-0600 h) after 45 h of wakefulness had little recuperative value relative to a nap taken during the rising phase of the cycle (1200-1400 h) after 53 h of wakefulness. A study of a 3-h nap in the nadir also showed little benefit from the sleep, prompting the conclusion that naps in or near the nadir may, for a number of reasons, lack recuperative power (23,24). Studies of performance after awakenings from varying amounts of recovery sleep (following sleep loss) also suggest that the first 1-2 h of sleep in the trough phase of the circadian cycle yields very poor performance (25-27). On the other hand, several studies have not reported a differential effect of naps due to circadian placement (11,14,28); of these, however, only one included a nap in the circadian nadir and this was prior to significant sleep loss (11). Thus, the extent to which alertness is affected as a function of the temporal placement of a nap within the circadian cycle and across days of otherwise sustained wakefulness remains unresolved, despite an abundance of nap studies.
When a nap has been shown to affect alertness, the effect varied with both the type
315 TEMPORAL PLACEMENT OF NAPS FOR ALERTNESS
of measure and the time of measurement. Thus, the question is: In what way and for how long is a nap likely to be beneficial as a function of its temporal placement? With regard to types of measure, performance was more often reported to be enhanced than was mood following naps during sustained wakefulness (11,14,23,24), although both were found to be improved in military field studies of naps (6,8).
Ironically, studies of naps as part of the normal sleep-wake cycle (i.e., without sustained wakefulness), have more often found the opposite, namely, enhanced mood without concomitant changes in performance (1,29-31), although Taub and colleagues reported improvement on both types of measure (28,32-34). Among the performance measures used in studies of sustained wakefulness, reaction time most often shows the best sensitivity to nap effects (8,11,14,22,35), but this may vary depending on the time that the measure is obtained after the nap.
Measurements taken within a few min of awakening from a nap typically show the negative impact of "sleep inertia" (4). Although sleep inertia is generally considered to dissipate within 30 min of daytime nap offset (36), Naitoh (22) suggested that it may be particularly prolonged when a nap is taken in the circadian nadir. A study using the Multiple Sleep Latency Test (MSLT) as a measure of alertness showed that the positive effects of a nap were not discernible until 2-4 h after the nap (13), but whether sleep inertia was the cause was uncertain. In general, once the period of sleep inertia is passed, positive effects of naps on performance are typically observed between 0.5 and 24 h after the nap, within the first 2 days of otherwise sustained wakefulness. When performance is assessed more distal from a nap (›or = 24 h), in the circadian nadir, the benefits of a nap have not been observed (14,37).
The issue of nap effects during sustained wakefulness is further complicated by the fact that severely sleep-deprived subjects may show considerable improvement on the number of items attempted in a cognitive task when told that a nap opportunity is imminent or that the experiment is coming to an end (38). This kind of motivated improvement is likely to be transient and to appear on subject-paced rather than experimenter-paced tasks. It suggests that subjects be told before a sleep-loss, protocol that a nap opportunity will be provided at some time. Even with such precautions, however, nap-benefit assessments conducted at the end of a sleep loss protocol may be contaminated by a transient motivational increment engendered by subjects' knowledge that the experimental trial is about to end.
In summary, it is not known to what extent the duration of nap benefits for alertness may vary with temporal placement of a nap both within a day and between consecutive days of sustained wakefulness, and what the post-nap time course of these benefits might be, relative to pre-nap alertness, baseline alertness, and no-nap alertness. Accordingly, we conducted a study for the purpose of assessing the effects on performance, subjective sleepiness and temperature cycles of a 2-h nap scheduled at opposite phases of the circadian cycle and after varying amounts of prior sleep loss.
METHODS
Design
The study consisted of five conditions that differed only in temporal placement of a 2-h nap opportunity during a 56-h period of otherwise sustained wakefulness in a controlled laboratory environment; subjects were randomly assigned to one of the five conditions. For a given condition, a nap could occur near either the circadian peak (P
316 D. F. DINGES ET AL.
= approximately 1500 h) or near the trough (T = approximately 0300 h) in the activity cycle. The P and T designates were based on sidereal time rather than on subjects' actual circadian cycles. Post-hoc analyses revealed, however, that the P or T designate for the phase timing of the nap was correct for each subject; that is, P naps always occurred a few hours prior to the peak of subjects' circadian temperature cycles, whereas T naps occurred a few hours before subjects' circadian nadir in temperature.
The three P-nap and two T-nap conditions differed in the amount of cumulative wakefulness prior to the nap. Pre-nap wakefulness was measured from the time subjects awoke from nocturnal sleep on the morning of the first day of the 56-h protocol; this was scheduled and confirmed to be at about 0900 h. The earliest nap condition was scheduled at 1500 h of the first day, 6 h after the morning awakening, and hence was designated as the P6 condition. A second condition involved a nap opportunity 12 h later, at 0300 h, after 18 h of prior wakefulness (T18 condition). A third condition provided a nap 12 h after T18 and 30 h after nocturnal sleep, at P30 (the afternoon of the second day). A fourth condition provided a T nap opportunity 12 h after P30 and after 42 h of continuous wakefulness (T42 condition), that is, during the second night of sleep loss. A fifth condition served as the control, permitting a nap 12 h after T42, near the end of the protocol, at P54. Thus, from 6 h of wakefulness onward, every 12 h throughout the protocol a nap opportunity was provided to one of the groups: P6, T18, P30, T42, or P54. The P6 and T18 naps took place prior to the first day of sleep loss and hence made operational the concept of a "prophylactic nap (20)." To study changes in sleep infrastructure and sleep inertia, these two conditions also included a second nap opportunity at P54, just before the sleep loss protocol ended, but these data are not relevant to this article. The T42 and P54 conditions correspond closely to Naitoh's (22) two nap conditions.
Subjects
Forty-one healthy young adults between the ages of 18 and 30 (mean age 20.4 years, SD 2.7) participated in the study. They were selected from a large population of university students based on their health, willingness to volunteer, and availability for sleep deprivation studies. All subjects were screened in advance to ensure that they had no medical or emotional problems and no sleep disorders, that they napped at least once a week, and that they had stayed awake in the past for ›or = 24 h. The reasons for these latter two requirements were to ensure that all subjects had some familiarity with both napping and sleep loss to prevent novelty of experience from increasing measurement error in the study; other data that we have collected over the years suggest that most students have experienced each, and there is no reason to believe that the results do not generalize beyond these requirements.
Assignment to nap condition was random, with an effort being made to equate the number of male and female subjects in each group. Table 1 presents the basic characteristics of subjects assigned to each of the five nap conditions. No significant differences were found among groups on any of these parameters, although fewer women than men volunteered for the study, resulting in fewer women subjects in three of the groups. The data from a tenth subject assigned to the P54 condition were eliminated from analyses after it was learned that he had ingested alcohol just prior to the sleep loss segment. The remaining subjects did not ingest alcohol or drugs prior to or during the 56-h trial in the laboratory. Subjects were run through the trial in same-sex pairs; they received a token reimbursement of $150 for participating, and they were informed
317 TEMPORAL PLACEMENT OF NAPS FOR ALERTNESS

that they could withdraw from the study at any time without jeopardy. An extensive debriefing interview was conducted at the end of the experiment, and follow-up contact was maintained with each participant. All subjects completed the study and encountered no problems after a full night of recovery sleep.
Protocol
Subjects underwent two baseline sessions, each 5 h in duration, during the 2 weeks prior to the 56-h period of sustained wakefulness in which the naps occurred. These sessions served to acquaint them with the study procedures, to provide practice sessions for the performance tasks, and to ensure that they maintained their usual sleep/ wake cycles during the study. The cycles were also checked by having subjects complete a daily sleep diary during the weeks prior to the sleep deprivation session.
Activities throughout the 56-h period of sustained wakefulness during which the naps occurred were structured so that only the placement of naps differed among groups. The environment was free of time cues (e.g., windows were occluded, and there were no clocks, televisions, radios, or telephones, etc.). Subjects were monitored continuously by behavioral observation. Performance test bouts that lasted approximately 45-60 min and included mood scale assessments and sublingual temperature recordings were conducted every 2.4 h throughout the 56-h trial. Between test bouts, subjects were free to read, play games, and engage in conversation, but not to sleep.
Meals took place four times a day, at times appropriate for breakfast, lunch, supper, and a midnight snack; the foods offered were of the type normally consumed at these times. Although the timing and content of the meals provided clear temporal cues (e.g., that it was morning, or evening, or the middle of the night), this was not considered to have compromised the time-free aspect of the protocol, since healthy young adults can readily predict during a 2- to 3-day period such broad variations in time with no cues at all. Most subjects cannot predict the precise hour of the day, however, and the meals were timed so that the hour was unclear. A few subjects actually wondered aloud whether the timing and content of the meals were being manipulated by the experimenters to create a false sense of time. This kind of concern was not due to any increase in paranoia resulting from sleep loss (since it was often mentioned early in the protocol), nor was it associated with a tendency to lose track of time (since subjects could readily predict the time, plus or minus 2 h, before a meal was served). Rather, it reflected subjects' inherent desire to guess the "true" reason for the experiment. Such concern for the meaning of any aspect of time-isolation, even if it is incidental to a
318 D. F. DINGES ET AL.
protocol, highlights the importance of demand characteristics (39) in determining subjects' responses to the protocol.
Debriefing interviews revealed that most subjects felt that the intent of the study was to measure response to sleep loss or temporal isolation or both. The between-subject design, together with staff discretion, prevented subjects from knowing that other nap conditions were also being tested. Consequently, the debriefing interviews determined that very few subjects guessed the intent of the experiment, which suggests that the results to be reported were not due to a differential motivation or response to demand characteristics.
Subjects knew that at some time during the 56-h trial a nap would be permitted, but they did not know when it would occur or how long it would last; they were told that its duration could vary from 10 min to 5 h. In fact, they were awakened 2 h after being told to go to sleep. The naps took place on beds in dark, quiet bedrooms. Sleep was polygraphically recorded and scored by standardized criteria. The effects of temporal placement on nap sleep psychophysiology have been reported elsewhere (40) and will be discussed here only to the extent that they shed light on the effects of the naps on other measures.
Performance
The test bouts included a variety of short-duration (1-20 min) cognitive and psychomotor tasks. These were administered in a predetermined random order by an experimenter who was present with the subject throughout the test. Subjects were continually urged to do their very best on each task. This instruction, the brevity of the tasks, their random placement in the bout, and the presence of the experimenter were intended to keep subjects' motivation to perform at the highest possible level while in the test room.
Although some cognitive tasks were administered in alternate test bouts, a simple, 10-min, unprepared visual reaction time (RT) task (41) was administered in every test bout. This task consistently has been shown to be sensitive to as little as one night's sleep loss (41,42), to partial sleep loss (19,43), and to circadian variation in performance efficiency (44,45). It was used as the performance measure of alertness in this report.
RT data were recorded as l-kHz frequency envelopes on audiotape in the portable device. A special microcomputer software system was developed (46) and used for inputting, editing, transforming, reducing, and analyzing the data. The system checks for and retrieves distorted data due to battery fluctuations and calculates variables associated with intratrial optimum response capacity (47), response slowing (48), and lapsing (49). The temporal patterns for the mean of the shortest 10% and the longest 10% of RT responses, as well as the median response within each trial were derived for each of the five groups.
Because RT data often result in a proportionality between the mean and SD, a reciprocal transformation was applied to the raw data (50). This has the effect of substantially decreasing the contribution of very long lapses (i.e., >2 s) and emphasizing slowing in the optimum and intermediate range of responses. The transformed data from each trial in the 56-h period were then expressed as a percentage of the equivalent variable in the final baseline trial and compared at each test-bout time between each of the four nap groups (P6, T18, P30, T42) and the control group (P54), using nonparametric statistical analyses.
319 TEMPORAL PLACEMENT OF NAPS FOR ALERTNESS
Sleepiness and temperature
Subjects completed the Stanford Sleepiness Scale (SSS) (51) while sublingual temperature was recorded at the beginning (pretest) and end (posttest) of each performance bout. Because performance bouts can influence the pattern of sleepiness ratings during sleep loss (52), between-group comparisons of temporal patterns in SSS as well as in temperature data were made separately for intertrial functions derived from pretest measures and those derived from posttest measures. The large number of tied scores necessitated parametric tests to compare SSS functions between groups. SSS ratings and temperature readings taken immediately when subjects awakened from naps were not included in these analyses.
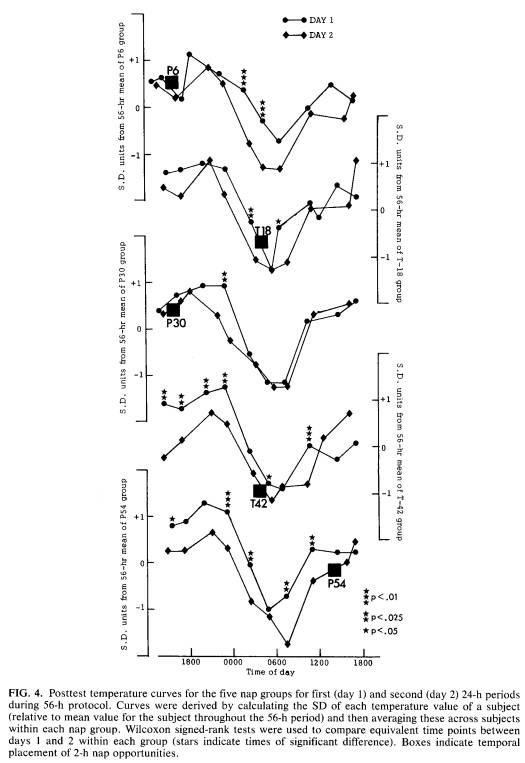
Posttest temperature curves for each 24-h period were compared within each group to determine if the naps systematically altered temperature. This was accomplished by replotting each subject's temperature cycle as a function of SD units from the mean of posttest temperature for the entire 56-h trial. In this way, temperature curves could be averaged across subjects within each nap group, permitting nonparametric signedranks tests to be made between the first and second 24-h periods of the protocol for each test-bout point comprising the circadian functions.
RESULTS
If napping improves alertness as measured by RT performance parameters and sleepiness ratings, these measures should be significantly different for a period of time after each nap condition when compared with the equivalent measures for the P54 control condition. There should be no differences between treatment groups and the control group prior to each nap.
RT performance
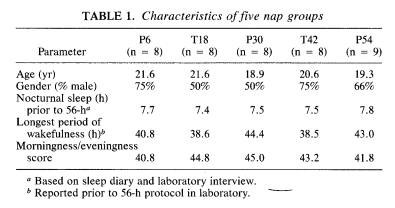
Lapse domain. Figure 1 shows the performance plots for the slowest 10% RTs (lapse domain) in each trial. Data from each of the four nap treatment groups are graphically compared with the P54 control group data. The most notable feature of the graphs is that all groups eventually displayed deterioration in performance capability (i.e., increased lapsing) as wakefulness increased beyond 24 h, even if a nap was taken. In all cases, the deterioration was moderated by the expected circadian pattern.
Naps significantly affected RTs in the lapse domain, however. The results of MannWhitney U tests at each trial test bout between the control group and respective treatment groups revealed that naps generally improved performance or prevented it from deteriorating as dramatically as it did with no nap opportunity. Of 35 comparisons made between treatment groups and the control group for trials after the naps, 23 (66%) produced significant differences. Of 25 comparisons made between groups for trials prior to the naps, 1 (4%) yielded significant results. Nap groups virtually never differed from the control group until after a nap was taken.
Although the positive effects of the P6 nap on RT lapses were not evident until 12 h after that nap (approximately 0500 h), when the P54 group had been awake for >20 h, these effects remained evident for most of the subsequent 30 h of the sleep loss trial (Fig. 1, upper left quadrant). This same result was found for the T18 nap, except that the differences appeared immediately after that nap (approximately 0500 h) and were even more robust (Fig. 1, upper right quadrant). The P30 nap also yielded significantly improved performance,
320 F. DINGES ET AL.
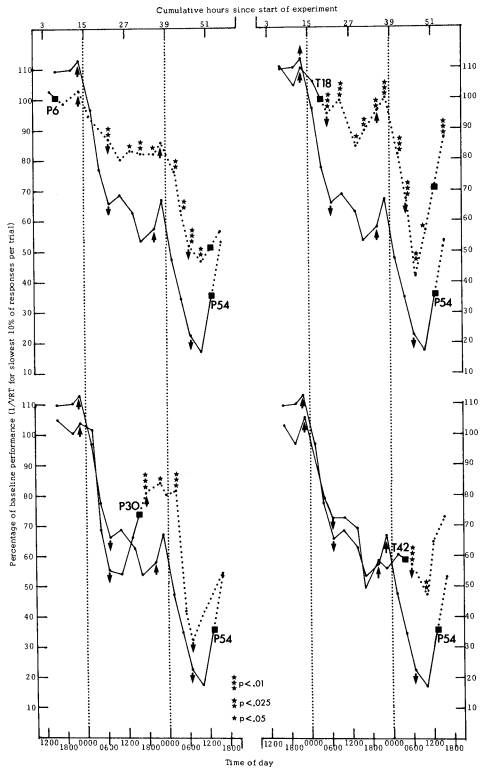
FIG. 1. Mean reaction time (RT) performance in lapse domain (slowest 10% of responses in each 10-min visual RT trial) for each of four nap treatment groups as compared with control nap group: Upper left, P6 versus P54; upper right, T18 versus P54; lower left, P30 versus P54; lower right, T42 versus P54. In all cases, data were transformed and expressed as a percentage of baseline performance (see text). Boxes indicate the 2-h nap opportunities. Dotted functions connect test points after treatment naps when differences from the control group were predicted. Test points that resulted in statistically significant Mann-Whitney U-test results are indicated with stars for level of significance. Upward arrows indicate average time of sublingual temperature peaks in each 24-h period; downward arrows indicate times of circadian nadirs.
321 TEMPORAL PLACEMENT OF NAPS FOR ALERTNESS
but only for approximately 8 h (Fig. 1, lower left quadrant). A similar result was observed following the T42 nap (Fig. 1 lower right quadrant).
Response slowing. Figure 2 shows comparisons between groups for the median RT value in each trial, which is distinct from both the lapse domain and the optimum response domain. This measure also decreased in all groups as a function of sleep loss, with the effect being most profound in the P54 group. Naps generally prevented some of the performance slowing observed in the control group, but half as many betweengroup differences were significant than were found in the lapse domain data; 12 of 35 (34%) comparisons made after naps were statistically significant versus no significant differences observed in 25 comparisons made prior to naps. As in the lapse domain, the bulk of the differences was observed for the P6 and T18 naps, particularly the latter (Fig. 2, upper right quadrant).
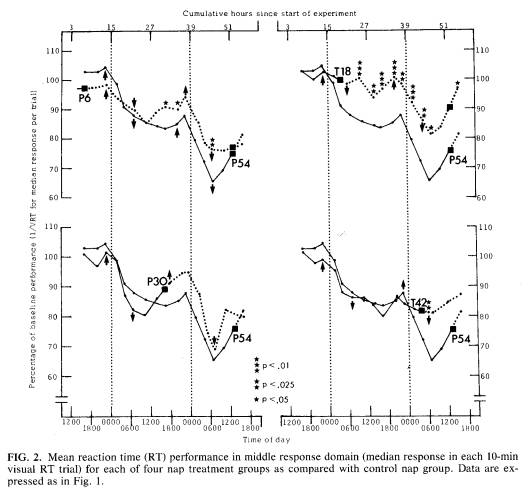
Optimum responses. The fastest 10% of RTs within a trial are shown in Fig. 3. Because of the transformation and analyses used, it was possible to show that sleep loss had an effect on this RT variable as well. Although the decline was modest, 39 of 41 subjects showed a loss of optimum response capability through the circadian nadir of the second night (approximately 0200-1000), and the effect was especially pronounced in the P54 group, who had not yet napped. Nap effects on the fastest 10% RTs were very modest; only 6 of 35 (17%) post-nap comparisons between groups were statistically significant compared to none of the 25 pre-nap comparisons. With one exception, the differences were confined to comparisons between the T18 and P54 groups.
Subjective sleepiness
Pretest SSS. Sleepiness ratings at the beginning of each performance bout increased significantly in all groups as the duration of wakefulness increased. Naps had no clear positive effect on pretest SSS ratings; only 4 of 35 (11%) between-group comparisons made after naps were statistically significant in the expected direction. The number of unexpected between-group differences found for comparisons made prior to naps was similar, however (4 of 29).
Posttest SSS. The results for posttest SSS ratings were no different from those of pretest ratings. Sleep deprivation was evident in all groups, but naps resulted in few differences (5 of 35 comparisons) and there were unexpected differences prior to naps (3 of 29 comparisons). The SSS differences that existed were not confined to any one nap group relative to the control group.
Temperature
Although all subjects showed circadian variation in oral temperature, posttest functions were less noisy than pretest functions owing to the constant posture and limited body movement during the preceding 45-min test interval. There were no clear differences between groups in either average pretest or posttest temperature functions.
322 F. DINGES ET AL.
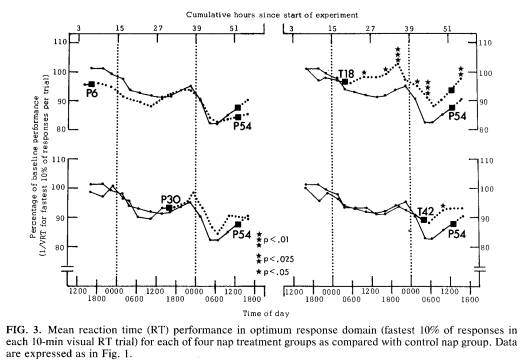
Posttest temperature cycles were assessed within each group to determine if the nap resulted in a mean change in temperature. Figure 4 shows the posttest temperature functions as variation around the 56-h mean for the first 24-h period (day 1) and for the second 24-h period (day 2) of the protocol within each group. The two functions differed significantly most often within the T42 and P54 groups. When a nap was not taken until late in the protocol (after 42 or 54 h), posttest temperature was likely to be lower at equivalent time points on day 2 relative to day 1; statistically significant differences were found for 11 of 20 comparisons (55%). The number is higher when only the comparisons made prior to the T42 and P54 naps (9 of 13 = 69%) are considered. Naps taken between 6 and 30 h of wakefulness resulted in considerably fewer posttest temperature comparisons that were significantly lower on day 2 (5 of 30, 17%).
DISCUSSION
A single 2-h nap placed anywhere during a 56-h period of sustained wakefulness improved performance on an RT task sensitive to lowered alertness. The improvement or, more accurately, the attenuation of performance decline, was not adequate to re
323 TEMPORAL PLACEMENT OF NAPS FOR ALERTNESS
store performance to baseline levels; clearly, more sleep would be required for this. Consistent with other studies of napping during prolonged wakefulness, the beneficial effects of the naps did not extend to self-report of mood (11,14,23,24); subjective sleepiness was not improved even transiently by only 2 h of sleep during 56 h. If the performance effects had been associated with differential incentive on the part of nap groups to appear more alert in accordance with the hypothesis, subjective self-report measures such as the SSS should also have evidenced benefits from the naps. Clearly, subjects were not responding to implicit experimental demand characteristics that may have existed. Rather, they seemed to be phenomenally unaware of their improved performance.
Subjective sleepiness is therefore a less sensitive index of nap effects in sustained wakefulness scenarios, but why this should be remains unclear; subjective sleepiness may be fundamentally different from other behavioral expressions of sleepiness (53) or may be more profoundly influenced by contextual variables (54,55) or more sensitive to sleep loss (than performance). Although changes in the SSS ratings of sleepiness may require more sleep than the 2-h nap opportunities provided, the lack of nap effects on SSS ratings cannot be attributed to the subjective ratings reaching maximum levels early in the protocol (averages for each group on the 7-point SSS ranged from 1.9 to 5.8 throughout the 56-h protocol). Indeed, there is no evidence that the SSS ratings were changing more dramatically or rapidly than was RT performance in the lapse domain (slowest 10%); the caveat here is that this is an impressionistic comparison since a different metric was used for the latter than for the former. SSS reports, however, indexed sleep loss and circadian variation more than the consequences of a nap, no matter where it was placed in the 56-h trial.
324 D. F. DINGES ET AL.
325 TEMPORAL PLACEMENT OF NAPS FOR ALERTNESS
The effect of a nap on RT performance was greatest for naps taken during the first 18 h, prior to the accumulation of significant sleep debt. It has been suggested that such "prophylactic" naps should increase alertness (20), but whether the benefits accrued would exceed those derived from napping after sleep loss accumulation was not known. This was the case, however; from approximately 0500 h of the first night, RT performance deteriorated less rapidly after the P6 and T18 naps. The P6 nap benefits were not evident for approximately 12 h. This delayed effect of a nap has also been observed in a study using the MSLT as a measure of alertness (13).
Differences between the prophylactic nap groups and the control group in the RT lapse domain were substantial and held for >24 h, through the second night. In contrast, effects from the P30 and T42 naps were less robust (i.e., fewer differences in all aspects of RT performance) and shorter lived. The benefits of the early naps were not simply an artifact of the P54 group being different from the P6 and T18 groups in response to sleep loss. When the P6, T18, and P30 nap groups were compared with the T42 group, the same relative pattern of results emerged. Improved performance was evident after each of the naps, especially the early naps, until the T42 nap. Although the T18 group seemed to benefit most from their nap relative to all other nap conditions, RT performance after T18 was not significantly above performance levels after other nap conditions when assessed at comparable time points.
The performance gains from the early naps are surprising when considered together with the sleep infrastructure of the naps (40). Subjects were not as sleepy prior to the P6 and T18 naps (stage 1 latency averaged 9.8 min and 4.8 min, respectively) as they were prior to the later naps (P30, T42, and P54 groups each averaged sleep latencies under 2 min). The P6 and T18 prophylactic naps also involved qualitatively "lighter" sleep than did later naps (cf. 40). They were significantly shorter (total sleep time = 80 and 106 min, respectively) than the other three nap conditions (each averaged 117 min), and they contained on the average significantly less slow wave sleep (SWS) (P6 = 14.6 min, T18 = 47.5 min) than did the later naps by the other groups (P30 = 63.3 min, T42 = 73.9 min, P54 = 72.0 min). In terms of relative performance benefits, therefore, the depth of the sleep after prolonged prior wakefulness was considerably less important than simply being able to obtain sleep prior to prolonged wakefulness.
This finding is unexpected if for no other reason than it is usually assumed that "deeper" sleep is likely to be more beneficial for subsequent wake functioning than "lighter" sleep. This may be generally true except in those cases in which the sleep loss exceeds 24 h and the "recovery" sleep is relatively brief (e.g., a 2-h nap). The sleep (nap) is inadequate to "pay-off" the sleep "debt," and the intensity of the sleep (as assessed by SWS latency and amount) is such that it may result in residual carryover (i.e., prolonged sleep inertia) into wake functioning. Whether this explanation accounts for the results of the P30 and T42 naps, the present data indicate, contrary to what was assumed in numerous studies of napping during sustained work, that it is evidently more important to prevent sleep debt accumulation by napping early than to attempt to pay it off later by napping deeply.
Naps taken during the first 30 h of the protocol also appeared to prevent the lowering of sublingual temperature between days 1 and 2, which occurred more frequently in the two groups that napped late in the protocol. Although the number of test-bout points comprising these functions are too few to estimate whether this temperature decline involved a change in circadian phase, period, or amplitude, the result suggests that even a modest amount of sleep can attenuate the decline in mean temperature that
326 D. F. DINGES ET AL.
often occurs during the first few days of sleep loss (56). Unfortunately, the significance of this mean temperature drop across days of wakefulness for our understanding of sleep remains obscure.
Whereas the timing of naps across days was an important determinant of their effects on temperature and performance, circadian placement of the naps, at least at the opposite phases of the cycle studied here, did not relate in any way to the RT performance benefits of the naps. Specifically, the work of Naitoh and colleagues (22-24) suggested that the nap taken in the circadian nadir after 42 h of wakefulness (T42) would not result in performance benefits. It did, however, in the present study, although too few performance trials were administered after the P54 nap to permit comparisons between the benefits of the T42 nap relative to the benefits of the P54 nap. This latter problem may have contributed to the findings of Naitoh's study (22). Consistent with the present results, however, he reported that a 3-h nap in the circadian nadir (after 2 days of wakefulness) was beneficial for both Four-Choice and simple RT performance, but not for cognitive performance and mood reports (23). Although a profound sleep inertia existed immediately after all naps in the current study, the data from this period were not included in the data analyses, but will be presented in a separate article. Rather, data reported here were collected from measurement bouts conducted at least 20 min after subjects were awake, moving around and interacting; at this time, no evidence was apparent of the kind of disorientation and cognitive impairment found on abrupt awakening from naps and known to dissipate in 5-30 min (36). Because of the apparent transient nature of sleep inertia, how the concept can be used to explain performance from 2 to 12 h after a nap is unclear.
Although work continues on the cognitive performance tasks and various mood reports gathered in the present study, the analysis of simple RT performance has proven not only a robust measure of nap effects, but also a remarkably sensitive index of sleep loss (19,41-43). Although changes in the lapse domain and to a lesser extent in the median response domain are expected as sleep loss increases, this study demonstrates that even optimum performance capability on the task declines with prior wakefulness if performance is analyzed to address this domain.
The visual RT sensitivity to prior wakefulness and naps was probably not due to where it was placed within the 45-60 min performance bouts, since this was randomly determined within trials and between subjects. The irony of such a brief, simple task being sensitive to sleep loss is that by traditional evaluations of "ideal" performance measures it should be profoundly insensitive to sleep loss (41,57-59). This is especially true for the simple, unprepared, visual RT task of Wilkinson and Houghton (41) since it provides feedback of results to the subject, which should hold interest and counteract sleepiness effects. The results consistently obtained with this task as well as with the auditory version on which it is based (60), compel a reevaluation of the lapse hypothesis and its expression in behavioral performance (55). The expression of sleepiness in performance appears to involve more than overt behavioral lapses in responding on long (>30 min), boring (e.g., vigilance), complex tasks. Studies of "fragmented wakefulness" (61) and response habituation (48) suggest that the behavioral manifestations of sleepiness are often more subtle than the conspicuous effects accounted for by traditional formulations of the lapse hypothesis.
In terms of measuring sleepiness, its expression, especially in biobehavioral measures, appears to be contextually dependent (55). As long as short-duration performance tasks capitalize on this contextual dependence, just as long-duration vigilance
327 TEMPORAL PLACEMENT OF NAPS FOR ALERTNESS
tasks and sleep latency tests do, no a priori reason prevents their exploration as practical measures of alertness. The data on RT suggest that this and other short-duration, portable performance tasks should be studied to develop practical, reliable, and highly sensitive behavioral measures of alertness that will provide a much-needed functional dimension to the MSLT (62) and other sensitive psychophysiological indices of sleepiness. Using the MSLT for criterion validity, these measures may have the added advantage that they do not suffer from ceiling effects and yet remain responsive to small amounts of sleep (naps) during sustained work scenarios in ways not evident in subjective impressions of sleepiness.
Acknowledgment: This research was supported by U.S. Office of Naval Research contract No. N00014-80-C-0380 and in part by a grant from the Institute for Experimental Psychiatry Research Foundation. We gratefully acknowledge the help of Stephen R. Fairbrother, Chris Auxier, John W. Powell, Mary F. Auxier, Ann Maliniak Whitehouse, Christine M. Dinges, Richard Barras, Elizabeth Whitehouse, and Jeffrey Moore in conducting this study.
REFERENCES
1. Dinges DF, Orne MT, Orne EC, Evans FJ. Voluntary self-control of sleep to facilitate quasi-continuous performance. Fort Detrick, Frederick, MD: US Army Medical Research and Development Command. NTIS No. AD-A102264, 1980.
2. Johnson LC. Sleep deprivation and performance. In: Webb WB, ed. Biological rhythms, sleep, and performance. Chichester: Wiley, 1982:111-38.
3. Hartley LR. A comparison of continuous and distributed reduced sleep schedules. Q J Exp Psychol 1974;26:8-14.
4. Lubin A, Hord D, Tracy ML, Johnson LC. Effects of exercise, bedrest and napping on performance decrement during 40 hours. Psychophysiology 1976;13:334-9.
5. Mullaney DJ, Kripke DF, Fleck PA, Johnson LC. Sleep loss and nap effects on sustained continuous performance. Psychophysiology 1983;20:643-51.
6. Haslam DR. Sleep deprivation and naps. Behav Res Methods Instrum Comput 1985;17:46-54.
7. Angiboust R, Gouars M. Tentative d'evaluation de l'efficacite operationelle du personnel de l'aeronautique militaire au cours de veilles nocturnes. In: Colquhoun WP, ed. Aspects of human efficiency: diurnal rhythm and loss of sleep. London: English Universities Press, 1972:151-70.
8. Opstad PK, Ekanger R, Nummestad M, Raabe N. Performance, mood, and clinical symptoms in men exposed to prolonged, severe physical work and sleep deprivation. Aviat Space Environ Med 1978;49:1065-73.
9. Haslam DR. The military performance of soldiers in continuous operations. In: Johnson LC, Tepas DI, Colquhoun WP, Colligan MJ, eds. Biological rhythms, sleep and shift work. New York: SP Medical & Scientific Books, 1981:217-30.
10. Thorne D, Genser S, Sing H, Hegge E Plumbing human performance limits during 72 hours of high task load. In: Forshaw SE, ed. Proceedings of the 24th defense research group seminar on the human as a limiting element in military systems, vol 1. Toronto: NATO Defence Research Group, NATO-DRG Report No. DS-A-DR(83) 170, 1983:17-40.
11. Gillberg M. The effects of two alternative timings of a one-hour nap on early morning performance. Biol Psychol 1984;19:45-54.
12. Nicholson AN, Pascoe PA, Roehrs T, Roth T, Spencer MB, Stone BM, Zorick E Sustained performance with short evening and morning sleeps. Aviat Space Environ Med 1985;56:105-14.
13. Lumley M, Roehrs T, Zorick F, Lamphere J, Roth T. The alerting effects of naps in sleep-deprived subjects. Psychophysiology 1986;23:403-8.
14. Webb WB. The proximal effects of two and four hour naps within extended performance without sleep. (submitted for publication).
15. Morgan BB. Effects of continuous work and sleep loss in the reduction and recovery of work efficiency. Am Ind Hyg Assoc J 1974;35:13-20.
16. Alluisi EA, Coates GD, Morgan BB. Effects of temporal stressors on vigilance and information processing. In: Mackie RR, ed. Vigilance: theory, operational performance and physiological correlates. New York: Plenum Press, 1977:361-424.
17. Alluisi EA. Influence of work-rest scheduling and sleep loss on sustained performance. In: Colquhoun WP, ed. Aspects of human efficiency: diurnal rhythm and loss of sleep. London: English Universities Press, 1972:199-215.
328 D. F. DINGES ET AL.
18. Carskadon MA, Dement WC. Cumulative effects of sleep restriction on daytime sleepiness. Psychophysiology 1981;18:107-13.
19. Tilley AJ, Wilkinson RT. The effects of a restricted sleep regime on the composition of sleep and on performance. Psychophysiology 1984;21:406-12.
20. Evans FJ, Orne MT. Recovery from fatigue: report No. 65. Fort Detrick, Frederick, MD: US Army Medical Research and Development Command. NTIS No. AD-A100310, 1976.
21. Haslam DR. Sustained operations and military performance. Behav Res Methods Instrum Comput 1985;17:90-5.
22. Naitoh P. Circadian cycles and restorative power of naps. In: Johnson LC, Tepas DI, Colquhoun WP, Colligan MJ, eds. Biological rhythms, sleep and shift work. New York: SP Medical & Scientific Books, 1981:553-80.
23. Naitoh P, Englund CE, Ryman D. Restorative power of naps in designing continuous work schedules. J Hum Ergol (Tokyo) 1982;1l(suppl):259-78.
24. Englund CE, Ryman DH, Naitoh P, Hogdon JA. Cognitive performance during successive sustained physical work episodes. Behav Res Methods Instrum Comput 1985;17:75-85.
25. Aschoff J, Giedke H, Poppel E, Wever R. The influence of sleep-interruption and of sleep-deprivation on circadian rhythms in human performance. In: Colquhoun WP, ed. Aspects of human efficiency: diurnal rhythm and loss of sleep. London: English Universities Press, 1972:135-50.
26. Akerstedt T, Gillberg M. Effects of sleep duration on memory and sleep latencies in connection with repeated awakenings from sleep. Psychophysiology 1979;16:49-52.
27. Rosa RR, Bonnet MH, Warm JS. Recovery of performance during sleep following sleep deprivation. Psychophysiology 1983;20:152-9.
28. Taub JM. Effects of habitual variations in napping on psychomotor performance, memory and subjective states. Int J Neurosci 1979;9:97-112.
29. Evans FJ, Orne MT. Recovery from fatigue: report No. 60. Fort Detrick, Frederick, MD: US Army Medical Research and Development Command. NTIS No. AD-A100347, 1975.
30. Bertelson AD. Effects of napping and bedrest on performance and mood. Doctoral thesis, The Ohio State University, 1979.
31. Daiss SR, Bertelson AD, Ludy BT. Napping versus resting: effects on performance and mood. Psychophysiology 1986;23:82-8.
32. Taub JM, Tanguay PE, Clarkson D. Effects of daytime naps on performance and mood in a college student population. J Abnorm Psychol 1976;85:210-7.
33. Taub JM. Napping behavior, activation, and sleep function. Waking Sleeping 1977;1:281-90.
34. Taub JM. Effects of scheduled afternoon naps and bedrest on daytime alertness. Int J Neurosci 1982;16:107-27.
35. Godbout R, Montplaisir J. The performance of normal subjects on days with and days without naps. Sleep Res 1986;15:71.
36. Dinges DF, Orne EC, Evans FJ, Orne MT. Performance after naps in sleep-conducive and alerting environments. In: Johnson LC, Tepas DI, Colquhoun WP, Colligan MJ, eds. Biological rhythms, sleep and shift work. New York: SP Medical & Scientific Books, 1981:539-52.
37. Webb WB. Experiments on extended performance: Repetition, age, and limited sleep opportunities. Behav Res Methods Instrum Comput 1985;17:27-36.
38. Haslam DR. The incentive effect and sleep deprivation. Sleep 1983;6:362-8.
39. Orne MT. On the social psychology of the psychological experiment: with particular reference to demand characteristics and their implications. Am Psychol 1962;17:776-83.
40. Dinges DE Differential effects of prior wakefulness and circadian phase on nap sleep. Electroencephalogr Clin Neurophysiol 1986;64:224-7.
41. Wilkinson RT, Houghton D. Field test of arousal: a portable reaction timer with data storage. Hum Factors 1982;24:487-93.
42. Glenville M, Broughton R, Wing AM, Wilkinson RT. Effects of sleep deprivation on short duration performance measures compared to the Wilkinson Auditory Vigilance Task. Sleep 1978;1:169-76.
43. Herscovitch J, Broughton R. Performance deficits following short-term partial sleep deprivation and subsequent recovery oversleeping. Can J Psychol 1981;35:309-22.
44. Glenville M, Wilkinson RT. Portable devices for measuring performance in the field: the effects of sleep deprivation and night shift on the performance of computer operators. Ergonomics 1979;22:927-33.
45. Tilley AJ, Wilkinson RT, Warren PSG, Watson B, Drud M. The sleep and performance of shiftworkers. Hum Factors 1982;24:629-41.
46. Dinges DF, Powell JW. Microcomputer analyses of performance on a portable, simple visual RT task during sustained operations. Behav Res Methods Instrum Comput 1985;17:652-5.
47. Dinges DF, Orne MT, Orne EC. Does the lapse hypothesis account for all sleep deprivation effects? Paper presented at the 54th annual meeting of the Eastern Psychological Association, Philadelphia, PA, 1983.
48. Kjellberg A. Sleep deprivation and some aspects of performance: II. Lapses and other attentional effects. Waking Sleeping 1977;1:145-8.
329 TEMPORAL PLACEMENT OF NAPS FOR ALERTNESS
49. Williams HL, Lubin A, Goodnow JJ. Impaired performance with acute sleep loss. Psychol Monographs 1959;73: Whole No. 484.
50. Kirk RE. Experimental design: Procedures for the behavioral sciences. Belmont, California: Wadsworth, 1968.
51. Hoddes E, Zarcone V, Smythe H, Phillips R, Dement WC. Quantification of sleepiness: a new approach. Psychophysiology 1973;10:431-6.
52. Dinges DF, Orne MT, Orne EC. Sleepiness during sleep deprivation: the effects of performance demands and circadian phase. Sleep Res 1984;13:189.
53. Broughton R. Performance and evoked potential measures of various states of daytime sleepiness. Sleep 1982;5(suppl 2):S135-46.
54. Dement WC, Carskadon MA. Current perspectives on daytime sleepiness: the issues. Sleep 1982;5(suppl 2):S56-66.
55. Dinges DF The nature of sleepiness: causes, contexts, and consequences. In: Baum A, Stunkard AS, eds. Eating, Sleeping and Sex. New York: Plenum (in press).
56. Kleitman N. Sleep and wakefulness. Chicago: University of Chicago Press, 1963.
57. Johnson LC, Naitoh P. The operational consequences of sleep deprivation and sleep deficit. London: Technical Editing and Reproduction. NATO/AGARDograph No. 193, 1974.
58. Wilkinson RT. Sleep deprivation: performance tests for partial and selective sleep deprivation. In: Abt LE, Riess BF, eds. Progress in clinical psychology: Vol. 8. Dreams and dreaming. New York: Grune & Stratton, 1968:28-43.
59. Woodward DP. A user oriented review of the literature on the effects of sleep loss, work-rest schedules, and recovery on performance. Arlington, VA: Office of Naval Research Technical Report No. ACR-206, 1974.
60. Lisper H, Kjellberg A. Effects of a 24-hour sleep deprivation on rate of decrement in a 10-minute auditory reaction time task. J Exp Psychol 1972;96:287-90.
61. Valley V, Broughton R. The physiological (EEG) nature of drowsiness and its relation to performance deficits in narcoleptics. Electroencephalogr Clin Neurophysiol 1983;55:243-51.
62. Carskadon MA, Dement WC. The multiple sleep latency test: What does it measure? Sleep 1982;5(suppl 2):S67-72.